What Does It Mean to Have an Elevated D Dimer?
- Research
- Open Admission
- Published:
D-dimer as a biomarker for illness severity and mortality in COVID-nineteen patients: a case control report
Journal of Intensive Care volume viii, Article number:49 (2020) Cite this article
Abstract
Background
Over 5,488,000 cases of coronavirus disease-19 (COVID-19) have been reported since December 2019. We aim to explore risk factors associated with mortality in COVID-19 patients and appraise the use of D-dimer every bit a biomarker for disease severity and clinical outcome.
Methods
We retrospectively analyzed the clinical, laboratory, and radiological characteristics of 248 consecutive cases of COVID-19 in Renmin Infirmary of Wuhan University, Wuhan, People's republic of china from Jan 28 to March 08, 2020. Univariable and multivariable logistic regression methods were used to explore risk factors associated with in-hospital mortality. Correlations of D-dimer upon admission with disease severity and in-hospital mortality were analyzed. Receiver operating characteristic curve was used to determine the optimal cutoff level for D-dimer that discriminated those survivors versus non-survivors during hospitalization.
Results
Multivariable regression that showed D-dimer > ii.0 mg/L at access was the merely variable associated with increased odds of mortality [OR 10.17 (95% CI 1.10–94.38), P = 0.041]. D-dimer elevation (≥ 0.50 mg/L) was seen in 74.vi% (185/248) of the patients. Pulmonary embolism and deep vein thrombosis were ruled out in patients with high probability of thrombosis. D-dimer levels significantly increased with increasing severity of COVID-19 equally determined by clinical staging (Kendall'south tau-b = 0.374, P = 0.000) and breast CT staging (Kendall's tau-b = 0.378, P = 0.000). In-hospital mortality charge per unit was 6.9%. Median D-dimer level in non-survivors (due north = 17) was significantly higher than in survivors (north = 231) [6.21 (three.79–16.01) mg/L versus i.02 (0.47–2.66) mg/L, P = 0.000]. D-dimer level of > ii.fourteen mg/L predicted in-infirmary mortality with a sensitivity of 88.2% and specificity of 71.three% (AUC 0.85; 95% CI = 0.77–0.92).
Conclusions
D-dimer is usually elevated in patients with COVID-19. D-dimer levels correlate with disease severity and are a reliable prognostic marker for in-infirmary mortality in patients admitted for COVID-19.
Groundwork
Coronavirus disease-xix (COVID-19) is the illness caused by 2019-nCoV/SARS-CoV-2, a novel β coronavirus of grouping 2B [1]. The illness ranges from asymptomatic or mild infection to severe respiratory tract infections in humans such every bit those seen in astringent acute respiratory syndrome (SARS) and Heart Eastward respiratory syndrome (MERS). Presentations include fever, cough, dyspnea, watery diarrhea, myalgia, severe lymphopenia, prolonged coagulation profiles, cardiac affliction, and sudden death [2, three].
Since the emergence in Wuhan, Hubei province, China in December 2019, COVID-xix has increased rapidly in China and progressed worldwide. On January 30, 2020, WHO declared the outbreak equally a Public Health Emergency of International Concern (PHEIC). As of May 27, 5,488,825 cases take been confirmed globally, including in Americas, Europe, Eastern Mediterranean, S-East Asia, and Africa, and 349,095 deaths have been reported [4]. Coagulopathy was reported, and D-dimer elevations were seen in three.75–68.0% of the COVID-xix patients [ii, v, 6]. Previous studies in customs-acquired pneumonia (CAP) and chronic obstructive pulmonary disease (COPD) patients have shown that D-dimer level is higher in astringent cases and may be used as a prognostic biomarker [7,eight,9], and D-dimer > i μg/ml is one of the risk factors for mortality in adult inpatients with COVID-19 [6]. Withal, the role of D-dimer in COVID-19 patients has not been fully investigated. In this study, nosotros showed D-dimer levels in patient groups stratified by clinical severities, imaging staging, in-hospital death, and assessed the role of D-dimer equally a biomarker for affliction severity and clinical outcome.
Methods
Patients
We enrolled patients of confirmed COVID-nineteen referred to the Renmin Infirmary of Wuhan University (Wuhan, People's republic of china), a designated center prioritized in treating critical affliction, from January 28 to March 08, 2020. Confirmed cases were divers as those with epidemiological history, consistent with two clinical manifestations, and microbiological evidence (respiratory or blood specimens positive for SARS-CoV-2 by real-time opposite transcription polymerase chain reaction (RT-PCR) assay or virus gene sequencing) according to the Novel Coronavirus Pneumonia Diagnosis and Treatment Guideline (6th ed.) (in Chinese) published past the National Wellness Commission of China [ten]. Symptom onset is determined past the primeval clinical manifestations consistent with COVID-19, such equally fever, coughing, dyspnea, musculus hurting, diarrhea, and fatigue, recorded in medical history taken upon admission. Exclusion criteria included pregnancy, cancer, hematologic malignancy, chronic liver disease, astute coronary syndrome, surgery or trauma within thirty days, and patients without D-dimer testing upon access. We retrospectively collected demographic, clinical data, laboratory parameters, chest CT imaging, and prognosis through electronic nursing and medical records using standardized data collection class. This study was canonical by the institutional ideals lath of Renmin Hospital of Wuhan University (No. WDRY2020-K048).
Laboratory and imaging methods
Consummate blood count, coagulation profile, renal and liver function, creatine kinase, electrolytes, myocardial enzymes, CD4 and CD8 cell counts, C-reactive protein, and procalcitonin were collected routinely on admission. D-dimer level is tested using immunoturbidimetric analysis with reference range of 0–0.50 mg/Fifty (Sysmex, CS5100). Doppler ultrasound and CT pulmonary angiography were done for whatever patients with high clinical suspicion of pulmonary embolism/deep vein thrombosis (PE/DVT). Chest CT browse was done for all inpatients.
Severity assessment
Clinically, severity of the COVID-19 patients was classified into mild, moderate, severe, and critically ill according to the Novel Coronavirus Pneumonia Diagnosis and Treatment Guideline (6th ed.) past the National Health Commission of China (Supplement tabular array ane) [10]. Radiologically, the area of afflicted lungs consistent with viral pneumonia in each patient'southward first chest CT subsequently admission was measured and classified into ≤ 30%, 31–50%, and ≥ 50% of full lung area. According to oxygenation index (OI) at admission, patients were grouped into iv groups (group 1, OI ≥ 400 mmHg; group 2, OI 300–399 mmHg; group 3, OI 200–299 mmHg; group 4, OI < 200 mmHg). The scores of SOFA, qSOFA, ITSH for disseminated intravascular coagulation (DIC), Curb-65 for community-acquired pneumonia and Wells' rule [11], and the revised Geneva score [12] for assessing pulmonary embolism (PE) adventure for each patient were documented.
Statistics
Continuous data accorded with normal distribution and homogeneity of variance were expressed as mean ± SD and compared by independent samples t test or expressed every bit median (25–75th percentile) and compared by Wilcoxon rank sum test. Categorical variables were expressed equally number (percentage) and compared by Chi-foursquare tests or Fisher's exact test. To explore the risk factors associated with mortality, univariable and multivariable logistic regression models were used. Because the total number of deaths (northward = 17) in our study and to avert overfitting in the model, we excluded variables from the univariable assay if their between-grouping differences were not significant and if the number of events was as well small to calculate odds ratios. Therefore, we chose age, SOFA, qSOFA, ISTH-DIC score, Adjourn-65, lymphocyte count, and D-dimer as the seven variables for our multivariable logistic regression model. Correlations of D-dimer with clinical staging, breast CT staging, oxygenation index, and in-infirmary mortality were evaluated past Kendall's tau-b coefficient analysis. To assess the predictive value of D-dimer for mortality, receiver operating characteristic (ROC) analysis was conducted with calculations of the area under the ROC curve (AUC), sensitivity and specificity. Statistical analyses were performed with SPSS (v.22.0; SPSS Inc., Chicago, IL, USA), and P value less than 0.05 was considered statistically pregnant.
Results
Every bit a designated referral center for the novel coronavirus infection, all the patients hospitalized were confirmed with RT-PCR. Later excluding subjects using the exclusion criteria, we included 248 sequent inpatients between January 28 and March 8, 2020, in the final analysis. The mean historic period of the 248 patients was 63.0 ± 13.iv years, ranging from 27 to 88 years. The average time from symptom onset to admission was 11.5 ± 5.ane days. Well-nigh i third of the patients had comorbidities, with hypertension being the about mutual (31.five%), followed by diabetes mellitus (17.7%). Balmy to moderate cases, severe cases, and critically sick cases accounted for 36.3%, 43.5%, and twenty.2% of the patients, respectively. Average length of hospital stay was xxx.8 ± 12.iv days. Using the revised Geneva score, none belonged to the high probability group for risk of PE. 4 patients belonged to the high probability group using the Wells' rule. Fortunately, they were ruled out of PE/VTE by Doppler ultrasonography and CT pulmonary angiography. Seventeen patients died during hospitalization. D-dimer elevation (≥ 0.50 mg/L) was seen in 74.vi% (185/248) of the patients.
Since all patients with normal D-dimer (< 0.5 mg/L) at admission survived, patients were grouped into D-dimer levels of < ane, i–2, and > 2 mg/50 in the univariable and multivariable logistic regression models (Table ane). In univariable analysis, age, SOFA score, qSOFA score, ISTH-DIC score, CURB-65, lymphocytopenia, and elevated D-dimer were associated with death. When these variables were included in the multivariable logistic regression model, D-dimer greater than two mg/L at admission was the just variable associated with increased odds of mortality [OR 10.17 (95% CI i.10–94.38), P = 0.041].
The comparison of demographic and clinical characteristics between the normal D-dimer group and elevated D-dimer group are shown in Table 2. Major laboratory markers and breast imaging features upon admission were recorded (Tabular array 3). 35.5%, 31.0%, and 33.five% of the patients had affected lungs of ≤ 30%, 31–50%, and ≥ 50% of the total surface area. The predominant changes seen were basis glass opacity (54.0%), followed past patchy consolidation (21.4%), fibrous stripes (12.9%), and irregular consolidated nodules (11.7%). 67.7% of the patients received oxygen therapy, including nasal cannula/face mask (52.0%), not-invasive mechanical ventilation (x.5%), invasive mechanical ventilation (5.2%), and extracorporeal membrane oxygenation in i patient. Anticoagulation therapy was prescribed in 34.3% of the cases.
The distributions of D-dimer levels among patients with different clinical staging, chest CT staging, and who survived and deceased during hospitalization are presented in Figs. i, 2, 3, and iv. On admission, D-dimer levels significantly increased with increasing severity of COVID-19 as determined past clinical staging (Kendall's tau-b = 0.374, P = 0.000), chest CT staging (Kendall'south tau-b = 0.378, P = 0.000), and oxygenation index (Kendall's tau-b = 0.392, P = 0.000). Median D-dimer levels showed an about 7-fold increase from moderate to critically ill patients (4.76 [ii.02–13.thirty] mg/Fifty versus 0.vi [0.33–ane.49] mg/L, P = 0.000), a 5-fold increase from patients with ≤ 30% affected lung area to ≥ l% change (3.93 [1.28–12.31] mg/L versus 0.6 [0.33–1.42], P = 0.042), and an over ix-fold increase from oxygenation index groups 1 to 4 ( 6.17 [1.75–14.20] mg/L versus 0.64 [0.46–1.39] mg/L, P = 0.000). All of those who did non survive had increased D-dimer level upon admission. When compared between patients who survived and who died during hospitalization, a significantly higher D-dimer level was detected in non-survivors versus survivors (half-dozen.21 [iii.79–16.01] mg/50 versus 1.02 [0.47–ii.66] mg/L, P = 0047).
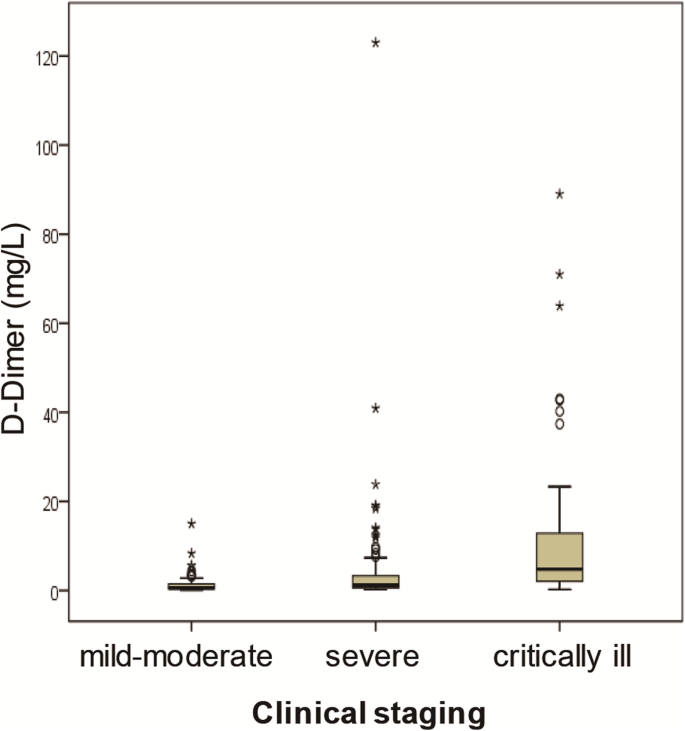
Correlations of D-dimer levels with clinical staging
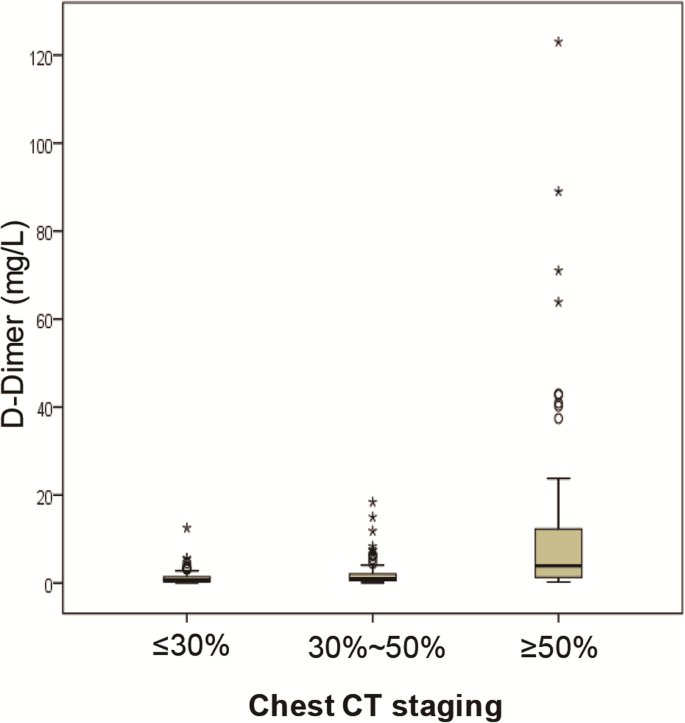
Correlations of D-dimer levels with chest CT staging according to area of afflicted lungs
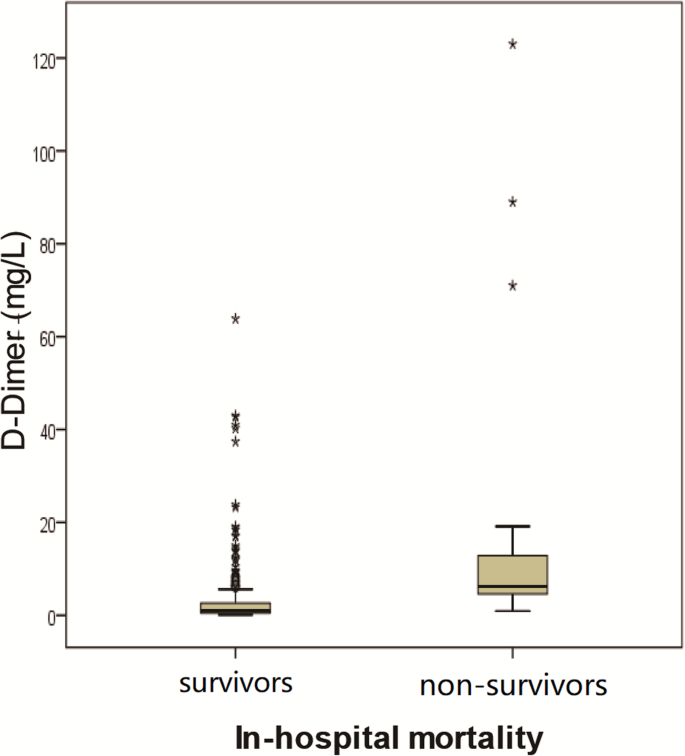
Correlations of D-dimer levels with in-hospital mortality
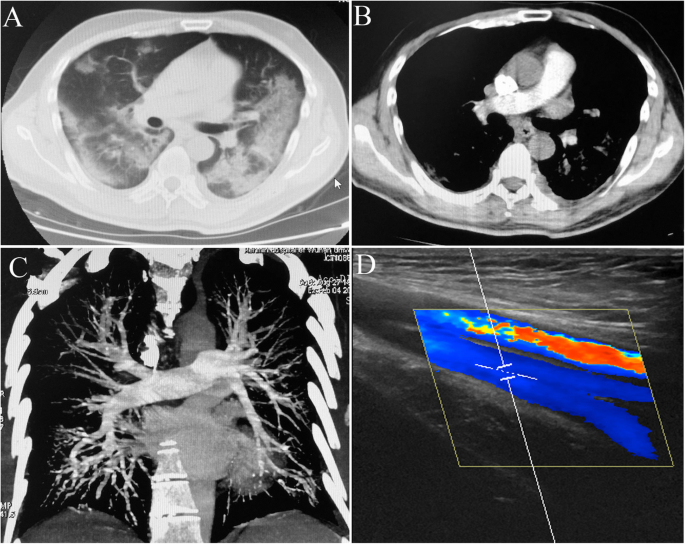
A 59-year-old male person diagnosed with COVID-xix who presented with fever, coughing, and hemoptysis. Chest CT upon admission showing basis glass opacities and patchy consolidation (a). He had an elevated D-dimer level of 9.43 mg/L. Wells' score, Geneva score, and CURB65 score were vii, 7, and 2 respectively. Wells' score suggested high probability of pulmonary embolism. CT pulmonary angiography (b, c) and Doppler ultrasonography (d) were and so carried out and ruled out pulmonary embolism and deep vein thrombosis in the lower extremities
ROC analysis identified D-dimer > 2.14 mg/50 upon admission as the optimal cutoff level to discriminate survivors from non-survivors (area under the ROC 0.85, standard error 0.037; 95% confidence interval [CI] 0.77–0.92, P = 0.000; Fig. 5). 32.7% of the included patients had a D-dimer of > 2.fourteen mg/L. For predicting in-hospital mortality, D-dimer level above ii.14 mg/Fifty had a sensitivity of 88.2% and specificity of 71.three% (Table 4).
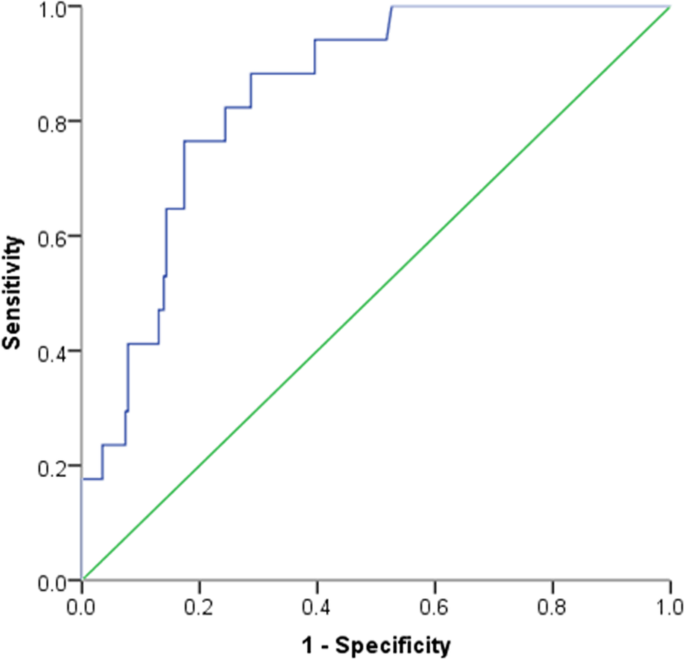
Receiver operating characteristic curve for D-dimer as parameter for predicting in-hospital mortality in COVID-19 patients
Give-and-take
We demonstrated that in patients diagnosed with COVID-nineteen, D-dimer elevation upon admission was mutual and was associated with both increased disease severity and in-hospital mortality. D-dimers are one of the fragments produced when plasmin cleaves fibrin to break down clots. The assays are routinely used equally part of a diagnostic algorithm to exclude the diagnosis of thrombosis. However, whatsoever pathologic or not-pathologic process that increases fibrin product or breakdown likewise increases plasma D-dimer levels [xiii]. Examples include deep vein thrombosis/pulmonary embolism, arterial thrombosis, disseminated intravascular coagulation, and conditions such as pregnancy, inflammation, cancer, chronic liver diseases, mail trauma and surgery status, and vasculitis. Amongst adults admitted to the emergency room, infections, instead of VTE/PE, are the most common reason for D-dimer elevation [14]. In the present study, no patient had confirmed PE/DVT, which supports the application of D-dimer in COVID-nineteen not just as a diagnostic tool for thromboembolism. In add-on, just three patients in the elevated D-dimer group (3/185, ane.vi%) with D-dimer levels of 42.8 mg/L, 89.0 mg/Fifty, and 71.0 mg/L had ISTH-DIC scores of ≥ 5, which is laboratory evidence compatible with overt DIC. Thus, the majority of the included patients with D-dimer peak in our study did not take overt DIC. Due to the retrospective nature of the study and pocket-sized number of patients with ISTH-DIC score consistent with overt DIC, it is hard to tell from our information if D-dimer tiptop is related with DIC.
Several studies have shown that D-dimer levels are associated with severity of community-acquired pneumonia and clinical outcome [seven, 15]. However, D-dimer has non been used every bit a biomarker for viral pneumonia [16, 17]. Though D-dimer summit has been observed in articles describing the clinical features of COVID-nineteen, whether the level of D-dimer is a marker of severity has not been examined.
In the nowadays study, there is a significant correlation between D-dimer levels and illness severity stratified by the surface area of afflicted lungs on chest CT, oxygenation index, also as clinical staging according to the interim guideline. In add-on, a college percentage of D-dimer peak was seen in the present study than previously reported [two, 5]. This may be due to the higher per centum of severe/critically ill cases referred to our infirmary, which is another demonstration of the correlation between D-dimer level and disease severity. This suggests that the assay may be used early as a marker of severity before chest CT scans or as a complement to CT and clinical staging.
In-hospital mortality was also associated with increased D-dimer levels, suggesting that the assay may exist used equally a single useful biomarker for clinical effect in patients with COVID-19. Zhou et al. reported that D-dimer > 1 μg/ml is a gamble for mortality [half-dozen]. The study objective, design, population, and statistical analysis of Zhou's report and those of ours are dissimilar. Zhou'due south study was a retrospective accomplice report to describe hazard factors for bloodshed and clinical grade, which included patients who had been discharged or had died by January 31, 2020. The mortality rate was higher compared to that in our study (28.3% vs. 6.9%). To explore risk factor for bloodshed, Zhou et al. chose age, coronary eye illness, SOFA score, lymphocyte, and D-dimer as variables for multivariable logistic regression model. D-dimer was divers every bit a chiselled variable in the assay, and levels of ≤ 0.five μg/L, > 0.five to ≤ 1 μg/L, and > ane μg/50 were chosen. The laboratory method for D-dimer assay was non described. In the nowadays instance control study, we focused on the predictive value of D-dimer for in-infirmary deaths using receiver operating characteristic analysis. In the assay, D-dimer is defined equally a continuous variable. Testing used immunoturbidimetric assay with reference range of 0–0.l mg/Fifty (Sysmex, CS5100). Despite the differences in study design and analysis, the findings and conclusions of the two studies are not inconsistent. Zhou et al. concluded that the potential risk factors of older historic period, high SOFA score, and D-dimer greater than one μg/L (instead of levels of ≤ 0.5 μg/L, or > 0.five to ≤ 1 μg/L) could help clinicians to place patients with poor prognosis. Nosotros found that when using the cutoff value of ii.14, D-dimer levels upon admission for in-hospital mortality has an AUC of 0.846. The sensitivity and specificity are 88.2% and 71.3%, respectively. The findings of this present study suggest that an elevated D-dimer level on admission (> ii.14 mg/50) may place patients at higher risk for in-infirmary bloodshed and therefore inform physicians about suitable candidates for intensive care and early on intervention.
It is worth noting that the findings suggest associations between D-dimer levels and disease severity and mortality only. Show is still defective every bit to the causal mechanisms and whether the associations are specific effects of SARS-CoV-2 infection or are consequences of systemic inflammatory response. In SARS-COV-two infection, dysregulation of coagulation/anti-coagulation cascades results in worsening lung pathology [18]. In influenza, the pathogenesis past augmenting viral replication and immune pathogenesis can be attributed to an abnormal coagulation organisation, including both the cellular and protein components [19]. The pathological features of COVID-19 include diffuse alveolar damage with cellular fibromyxoid exudates, desquamation of pneumocytes and hyaline membrane formation, pulmonary edema with hyaline membrane formation, and interstitial mononuclear inflammatory infiltrates, dominated by lymphocytes, which greatly resemble those seen in SARS and MERS coronavirus infection [20, 21]. Presumably, the observed D-dimer elevation signify a hyperfibrinolysis land and increased inflammatory burden induced in SARS-COV-ii infection. In our logistic regression model to estimate risk factors associated with mortality, systematic anticoagulation therapy was not significantly associated with reduced risk of mortality. Nonetheless, in a recent observational study including 2773 hospitalized COVID-19 patients, Paranjpe et al. found that treatment dose anticoagulant was associated with a reduced run a risk of mortality, especially among patients who required mechanical ventilation [22]. And longer duration of treatment was associated with a reduced hazard of bloodshed (adjusted Hr of 0.86 per day, 95% CI 0.82–0.89, p < 0.001). Whether anticoagulation therapy confers a survival benefit in patients hospitalized for COVID-19 needs farther inquiry with prospective randomized trials. Currently, the potential benefits need to exist weighed against risk of bleeding.
This study has some limitations. First, the current study was done in a single center. The overall mortality (vi.ix%) was lower compared with that reported in other studies done in Wuhan [2, half-dozen] and considerably higher than those reported by other provinces [5, 23]. Further researches may be needed when extrapolated to wider patient population. 2nd, the study is retrospective in nature. The patients included were not systematically assessed for the presence of PE/DVT simply but when clinically suspected. Third, we did non await into the value of series D-dimer monitoring in assessing COVID-xix patients.
Conclusions
In conclusion, D-dimer levels are normally elevated in patients infected with SARS-CoV-2. Significantly higher levels are constitute in those with critical disease and may be used as a prognostic mark for in-infirmary mortality.
Availability of data and materials
The dataset supporting the conclusions of this article is included within the article and its additional files.
Abbreviations
- COVID-19:
-
Coronavirus disease-xix
- SARS:
-
Astringent acute respiratory syndrome
- MERS:
-
Middle Due east respiratory syndrome
- PHEIC:
-
Public Wellness Emergency of International Concern
- CAP:
-
Community-acquired pneumonia
- COPD:
-
Chronic obstructive pulmonary affliction
- PE:
-
Pulmonary embolism
- DVT:
-
Deep vein thrombosis
- ISTH:
-
International Gild on Thrombosis and Haemostasis
- DIC:
-
Disseminated intravascular coagulation
References
-
Zhou P, Yang Ten, Wang X, Hu B, Zhang 50, Zhang Due west, Si H, Zhu Y, Li B, Huang C, et al. A pneumonia outbreak associated with a new coronavirus of likely bat origin. Nature. 2020;579(7798):270–iii.
-
Chen Northward, Zhou M, Dong X, Qu J, Gong F, Han Y, Qiu Y, Wang J, Liu Y, Wei Y, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive report. Lancet. 2020;395(10223):507–13.
-
Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, Zhang L, Fan One thousand, Xu J, Gu X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet (London, England). 2020;395(10223):497–506.
-
Coronavirus disease (COVID-19) State of affairs Report-128. https://www.who.int/emergencies/diseases/novel-coronavirus-2019/state of affairs-reports. (Accessed 28 May 2020).
-
Wu J, Liu J, Zhao Ten, Liu C, Wang Westward, Wang D, Xu Westward, Zhang C, Yu J, Jiang B,et al. Clinical Characteristics of Imported Cases of COVID-xix in Jiangsu Province: A Multicenter Descriptive Study. Clin Infect Dis. 2020;29:ciaa199.
-
Zhou F, Yu T, Du R, Fan Chiliad, Liu Y, Liu Z, Xiang J, Wang Y, Song B, Gu X, et al. Clinical course and gamble factors for bloodshed of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort written report. Lancet. 2020; 395(10229):1054-62.
-
Querol-Ribelles JM, Tenias JM, Grau Due east, Querol-Borras JM, Climent JL, Gomez E, Martinez I. Plasma d-dimer levels correlate with outcomes in patients with customs-acquired pneumonia. Chest. 2004;126(4):1087–92.
-
Fruchter O, Yigla K, Kramer MR. d-dimer as a prognostic biomarker for mortality in chronic obstructive pulmonary disease exacerbation. Am J Med Sci. 2015;349(1):29–35.
-
Snijders D, Schoorl M, Schoorl Grand, Bartels PC, van der Werf TS, Boersma WG. D-dimer levels in assessing severity and clinical outcome in patients with community-acquired pneumonia. A secondary assay of a randomised clinical trial. Eur J Intern Med. 2012;23(5):436–41.
-
Novel Coronavirus Pneumonia Diagnosis and Treatment Guideline (sixth ed.) (inChinese). 2020. http://www.nhc.gov.cn/xcs/zhengcwj/202002/8334a8326dd94d329df351d7da8aefc2.shtml. (Accessed 03 Mar 2020).
-
Wells P, Anderson D, Rodger K, Ginsberg J, Kearon C, Gent M, Turpie A, Bormanis J, Weitz J, Chamberlain M, et al. Derivation of a simple clinical model to categorize patients probability of pulmonary embolism: increasing the models utility with the SimpliRED D-dimer. Thromb Haemostasis. 2017;83(03):416–20.
-
Le Gal K, Righini Chiliad, Roy PM, Sanchez O, Aujesky D, Bounameaux H, Perrier A. Prediction of pulmonary embolism in the emergency department: the revised Geneva score. Ann Intern Med. 2006;144(3):165-71.
-
Linkins LA, Takach Lapner South. Review of D-dimer testing: good, bad, and ugly. Int J Lab Hematol. 2017;39(S1):98–103.
-
Lippi G, Bonfanti Fifty, Saccenti C, Cervellin M. Causes of elevated D-dimer in patients admitted to a large urban emergency department. Eur J Intern Med. 2014;25(ane):45–8.
-
Dai R, Kong Q, Mao B, Xu W, Tao R, Wang X, Kong Q, Xu J. The mortality risk factor of community acquired pneumonia patients with chronic obstructive pulmonary disease: a retrospective cohort written report. BMC Pulm Med. 2018;18(1):12.
-
Guo 50, Wei D, Zhang 10, Wu Y, Li Q, Zhou K, Qu J. Clinical features predicting mortality risk in patients with viral pneumonia: the MuLBSTA score. Front Microbiol. 2019;10:2752.
-
Yoon H, Jhun BW, Kim SJ, Kim K. Clinical characteristics and factors predicting respiratory failure in adenovirus pneumonia. Respirology. 2016;21(7):1243–l.
-
Gralinski LE, Baric RS. Molecular pathology of emerging coronavirus infections. J Pathol. 2015;235(ii):185–95.
-
Yang Y, Tang H. Aberrant coagulation causes a hyper-inflammatory response in astringent flu pneumonia. Cell Mol Immunol. 2016;13(iv):432–42.
-
Xu Z, Shi L, Wang Y, Zhang J, Huang L, Zhang C, Liu S, Zhao P, Liu H, Zhu L, et al. Pathological findings of COVID-nineteen associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;eight(4):420–2.
-
Channappanavar R, Perlman S. Pathogenic human coronavirus infections: causes and consequences of cytokine storm and immunopathology. Semin Immunopathol. 2017;39(5):529–39.
-
Paranjpe I, Fuster 5, Lala A, Russak A, Glicksberg BS, Levin MA, Charney AW, Narula J, Fayad ZA, Bagiella E, et al. Association of treatment dose anticoagulation with in-hospital survival among hospitalized patients with COVID-nineteen. J Am Coll Cardiol. 2020;76(ane):122–4.
-
Tian S, Hu North, Lou J, Chen K, Kang X, Xiang Z, Chen H, Wang D, Liu N, Liu D, et al. Characteristics of COVID-19 infection in Beijing. J Infect. 2020;80(4):401–6.
Acknowledgements
Not applicable
Writer information
Affiliations
Contributions
YY and JC contributed in the conception, design, acquisition and analysis, interpretation of data, and writing of the manuscript. QW, QS, KL, ZL, Xc, SC, and KY contributed in the pattern, conquering, and assay of information. ZH and BH contributed to the conception, design of the work, analysis of data, interpretation of data, and revision of the manuscript. All authors have approved the submitted version. All authors have agreed to be personally accountable for the author's own contributions and to ensure that questions related to the accurateness or integrity of whatever role of the work, even ones in which the author was non personally involved, are appropriately investigated and resolved and the resolution documented in the literature.
Corresponding authors
Ethics declarations
Ideals approval and consent to participate
This study was canonical past the institutional ethics board of Renmin Hospital of Wuhan University (No. WDRY2020-K048).
Consent for publication
Not applicative
Competing interests
The authors declare that they have no competing interests in this section.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution iv.0 International License, which permits use, sharing, accommodation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original writer(due south) and the source, provide a link to the Artistic Commons licence, and signal if changes were made. The images or other third political party fabric in this commodity are included in the article'southward Artistic Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the commodity's Artistic Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted utilize, you volition need to obtain permission straight from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/past/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data fabricated bachelor in this article, unless otherwise stated in a credit line to the data.
Reprints and Permissions
About this article
Cite this article
Yao, Y., Cao, J., Wang, Q. et al. D-dimer equally a biomarker for disease severity and mortality in COVID-nineteen patients: a instance control written report. j intensive care 8, 49 (2020). https://doi.org/x.1186/s40560-020-00466-z
-
Received:
-
Accustomed:
-
Published:
-
DOI : https://doi.org/x.1186/s40560-020-00466-z
Keywords
- D-dimer
- Coronavirus disease-xix
- Biomarker
- Severity
- Bloodshed
mccorkledepud1987.blogspot.com
Source: https://jintensivecare.biomedcentral.com/articles/10.1186/s40560-020-00466-z
Post a Comment for "What Does It Mean to Have an Elevated D Dimer?"